User Tools
Sidebar
Table of Contents
Chemical and Gas Adsorption on Nanostructures
Nanostructured materials, which ususally posses high surface/volume ratios, can be hosts of variouis adsorbates.
Aspirin on graphitic carbon structures
Using density functional theory, we investigate the adsorption properties of acetylsalicylic acid (aspirin) on the outer surfaces of a (10,0) carbon nanotube (CNT) and a (8,0) triazine-based graphitic carbon nitride nanotube (CNNT). The adsorption energies for the CNNT and CNT are 0.67 and 0.51 eV, respectively, and hence, the aspirin molecule binds more strongly to the CNNT. The stronger adsorption energy for the binding to the CNNT is ascribed to the high reactivity of its nitrogen atoms with high electron affinity. The CNNT exhibits local electric dipole moments that cause strong charge redistribution in the adsorbed aspirin molecule. The influence of an external electric field on the adsorption of aspirin on the nanotubes is explored by examining modifications in their electronic band structures, partial densities of states, and charge distributions. An electric field applied along a particular direction is found to induce molecular states of aspirin that lie within the in-gap region of the CNNT. This implies that the CNNT can be potentially utilized for the detection of aspirin.
Amino acids on graphitic carbon structures
Adsorption properties of Amino acids on graphitic carbon structures.
H2S detection in CNT-based devices
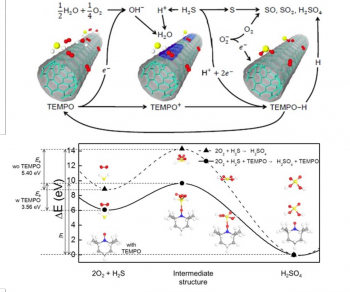 Here we report the highly effective detection of hydrogen sulfide
(H2S) gas by redox reactions based on single-walled carbon
nanotubes (SWCNTs) functionalized with
2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) as a catalyst and we also
discuss the important role of water vapor in the electrical
conductivity of SWCNTs during the sensing of H2S molecules.
To explore the H2S sensing mechanism, we investigate the
adsorption properties of H2S on carbon nanotubes (CNTs) and
the effects of the TEMPO functionalization using first-principles
density functional theory (DFT) and we summarize currentchanges of
devices resulting from the redox reactions in the presence of
H2S. The semiconducting-SWCNT (s-SWCNT) device
functionalized with TEMPO shows a very high sensitivity of 420% at 60%
humidity, which is 17 times higher than a bare s-SWCNT device under
dry conditions. Our results offer promising prospects for personal
safety and real-time monitoring of H2S gases with the
highest sensitivity and low power consumption and potentially at a low
cost.
Here we report the highly effective detection of hydrogen sulfide
(H2S) gas by redox reactions based on single-walled carbon
nanotubes (SWCNTs) functionalized with
2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) as a catalyst and we also
discuss the important role of water vapor in the electrical
conductivity of SWCNTs during the sensing of H2S molecules.
To explore the H2S sensing mechanism, we investigate the
adsorption properties of H2S on carbon nanotubes (CNTs) and
the effects of the TEMPO functionalization using first-principles
density functional theory (DFT) and we summarize currentchanges of
devices resulting from the redox reactions in the presence of
H2S. The semiconducting-SWCNT (s-SWCNT) device
functionalized with TEMPO shows a very high sensitivity of 420% at 60%
humidity, which is 17 times higher than a bare s-SWCNT device under
dry conditions. Our results offer promising prospects for personal
safety and real-time monitoring of H2S gases with the
highest sensitivity and low power consumption and potentially at a low
cost.
- High-performance H2S detection by redox reactions in semiconducting carbon nanotube-based devices
Hyun Young Jung, Young Lae Kim, Sora Park, Aniket A. Datar, Hyung-June Lee, Jun Huang, Sivasubramanian Somu, Ahmed Busnaina, Yung Joon Jung, and Young-Kyun Kwon, Analyst 138 (23), 7206-7211 (2013).
link: download:
download: 

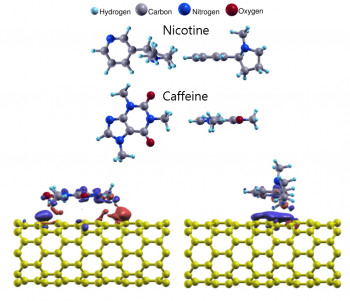
Nicotine and caffeine on CNTs
Using first-principles calculations, we investigate the electronic structures and binding properties of nicotine and caffeine adsorbed on single-walled carbon nanotubes to determine whether CNTs are appropriate for filtering or sensing nicotine and caffeine molecules. We find that caffeine adsorbs more strongly than nicotine. The different binding characteristics are discussed by analyzing the modification of the electronic structure of the molecule-adsorbed CNTs. We also calculate the quantum conductance of the CNTs in the presence of nicotine or caffeine adsorbates and demonstrate that the influence of caffeine is stronger than nicotine on the conductance of the host CNT.
- Molecular adsorption study of nicotine and caffeine on single-walled carbon nanotubes from first principles
Hyung-June Lee, Gunn Kim, and Young-Kyun Kwon, Chem. Phys. Lett. 580, 57-61 (2013).
link: download:
download: 

Metal adsorbates on graphene
Metal atoms on graphene, when ionized, can act as a point-charge impurity to probe a charge response of graphene with the Dirac cone band structure. To understand the microscopic physics of the metal-atom-induced charge and spin polarization in graphene, we present scanning tunneling spectroscopy Metal adsorbates on graphene STS simulations based on density-functional theory calculations. We find that a Cs atom on graphene is fully ionized with a significant band-bending feature in the STS whereas the charge and magnetic states of Ba and La atoms on graphene appear to be complicated due to orbital hybridization and Coulomb interaction. By applying external electric field, we observe changes in charge donations and spin magnetic moments of the metal adsorbates on graphene.
- Tunable charge donation and spin polarization of metal adsorbates on graphene using an applied electric field
Jae-Hyeon Parq, Jaejun Yu, Young-Kyun Kwon, and Gunn Kim, Phys. Rev. B 82 (19), 193406 (2010).
link: download:
download: 

Glycines on CNTs for alcohol sensing
Glycines are spontaneously adsorbed to form into self-assembled nanoclutsers on single-walled carbon nanotubes (SWNTs). After formation of glycine nanoclusters on SWNTs, the field effect transistor (FET) devices show selective sensing ability to alcohols, such as isopropyl alcohol (IPA), methanol, and ethanol. Upon the adsorption of alcohol, the glycine-coated SWNT-FET devices exhibit pseudo-metallic transport behaviors, whereas the original and glycine-coated devices display conventional p-type transport characteristics. Computational studies support that the gate field screening effect induced by instantly formed glycine-alcohol pair layers seems to be responsible for the pseudo-metallic transport behavior.
- Self-clusterized Glycines on Single Walled Carbon Nanotubes for Alcohol Sensing
Hyun Jae Song, Yoonmi Lee, Tao Jiang, Adil-Gerai Kussow, Minbaek Lee, Seunghun Hong, Young-Kyun Kwon, and Hee Cheul Choi, J. Phys. Chem. C 112 (2), 629-634 (2008).
link: download:
download: